The Nature of Electricity¶
- Reference1
- Reference2
Computers are electical gadgets. That should be obvious, since we need an electrical power source of some kind to make them do anything. If that power source goes away, a computer is a useless pile of physical stuff that can be used as a door-stop!
Once we add that power source, magic starts to happen.
Power Source?¶
We power our computers either with a battery, or by plugging it into a wall socket. In either case, that power source provides a pile of electrons that can move through our computer.
Electrons¶
So what is an electron?
This is not a Physics course, so we will not dig too deeply into electricity here. Basically, an electron is a tiny particle that carries a negative electrical charge. There are other kinds of particles in our universe, including protons, which carry a positive electrical charge (opposite from that carried by an electron), and neutrons, which carry no charge at all.
Atoms¶
Atoms are made up of these three fundamental particles. In general any material is made up of a bunch of atoms, each identical in structure:
Some number of neutrons
Some number of protons
Usually the same number of electrons as we have protons.
The neutrons and protons are glued together in a mass called the nucleus of the atom. The electrons swarm around that nucleus in a cloud. The best way to visualize this is to compare it to our solar system. The nucleus is the Sun, and the electrons are like the planets orbiting around the sun. The big difference is that electron orbits do not resemble a typical picture of our planets that much. Instead electrons live in one of several “shells” each of which contains a number of electrons all spinning around at some common speed. Their “orbits” are not nicely laid out like our planets are.
These shells are called electron shells. The outermost shell is called the valence shell, and that is where electrons live that can can interact with the electrons from other atoms if they get close enough together.
Here is a picture of a copper atom:
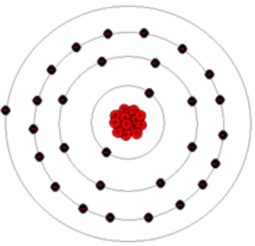
Copper atoms have this structure:
Core consisting of
34 neutrons
29 protons
Electron cloud with 29 electrons
The electrons we are interested in tend to hang out near those outer rings called valence rings, rather than in orbits closer to the center. It takes energy to move an electron from one ring to another, and more energy to knock an electron loose from an atom.
Free Electrons¶
Some atoms will tolerate extra electrons which can hang out on that outer ring. These electrons are not as tightly bound to the atom, and they can move around between atoms more easily. We call these “free electrons” and they are really what is moving around when we talk about electricity!
But the action is not what you might expect.
One analogy I saw was pretty interesting. Pretend that these free electrons are sitting in a tube like marbles packed together in that tube. The atoms are as close together as they can get. If we push one free electron into the end of the tube, one will need to pop out of the other end to make room. Just as would be the case if we were playing with marbles. (Wait, I seem to have lost a marble somewhere!)
We see an electron pop out at the other end as a result of pushing a new on in. All of the electrons in the tube only moved a small amount, but we see the action immediately on the other end. Wow! Cool stuff!
Clueless Atoms¶
Just as we discussed when talking about our digital gadgets, atoms have no clue what is going on around them. They just sit there watching over their private swarm of electrons. If a few free ones come by and seems to stick around, the atom is more than happy to pitch them away, so some other atom can deal with them.
This idea led me to wake up in the middle of the night with a game idea we can play to show how electricity flows.
Are you ready?
Let’s Dance!
